Issue 96
Covid-19 testing chaos, re-making existing drugs, all the Covid-19 vaccines in production

August 2, 2020
Hello friend! Welcome to Scrap Facts.
I'm a reporter covering health and science with insatiable curiosity. I love everything I learn, not all of which gets its own story. Each week, I'll bring you some of my favorite facts that I picked up on the job or while out living life.
Archives from Tinyletter can be found here. Love Scrap Facts? Consider hitting the “like” button, or tell your friends to sign up!
The reason Covid-19 tests take so long? There are too many kinds of them.
Found while reporting: Why Covid-19 test results are taking so long.
In the US, we had a massive shortage of Covid-19 tests back in February because the ones from the US Centers for Disease Control and Prevention were faulty. So the US Food and Drug Administration decided that the pandemic warranted the temporarily authorization of new tests. These didn’t have to go through the usual rigorous testing and evaluations, which would take a lot of time. The point was to get them out there.
Now there are ~150+ testing kits that can detect the presence of SARS-CoV-2 DNA. Hospital labs have ordered a hodgepodge of these testing kits—some from on company, some from another, and so on—just to be able to meet their testing demands.
They all do the roughly the same thing, but there are minor differences in the specific equipment they each need. If there’s a shortage of one type of hyper-specific plastic pipette, the whole machine or kit is put out of use, which puts even more strain on the other testing equipment in the lab.
Hospital labs are quickly overrun with Covid-19 test orders, which means they must outsource them to other hospitals or labs. Two of the biggest lab companies are LabCorp and Quest Diagnostics. Both of those groups boast being able to process between 135,000 and 180,000 tests per day. But they’ve also got their own tests to process—they take in tests from either local or state health departments or some of those pop-up testing clinics you may have seen.
So, even though tests should take a day or so for high-priority patients or a few days for low-priority folks, they end up taking two weeks or so. Which isn’t great—there’s a lot of time you could become exposed to the virus after getting your nasal or saliva swab.
The US government right now is giving millions of dollars in loans or grants to a handful of companies to find new ways to make drugs that already exist.
Found while reporting: The US wants to use Covid-19 to insource drug manufacturing.
The ballpark figure for how long it takes for a drug to go from research and development to the market is a decade. During that time, the company making it is spending millions (or even billions!) to bring it to market. Only about 1/3 of drugs in development actually make it to market.
All of this is to say, when drug companies find a formula that works, they don’t mess with it. Even if they could be making it more efficiently, with less waste or with fewer materials. Because drugs have to be chemically perfect (they’re dealing with human health, after all), it’s far too financially risky for a drug company to try to tinker with a recipe that works.
But if you could do it—find a better way to make exactly the same product—you could make things a lot cheaper. Which theoretically could mean that drugs could cost less (although the cost of making a drug is only loosely reflected in the price consumers pay). And, more importantly, the US could stop buying drugs from foreign companies. Which could be a measure of safety if supply chains were messed up because of a global pandemic (which they were), but it’s a bit nationalistic too.
So that’s why the US government has decided to invest in Kodak (a former photography company) and Phlow, a newly-created (as of 2020) drug company. The hope is that these companies will be able to make the manufacturing shifts that existing drug companies can’t.
And finally, a breakdown of all the statuses of the Covid-19 vaccines farthest along in development.
Found while reporting: Why the US’s vaccine investment isn’t purely driven by speed.
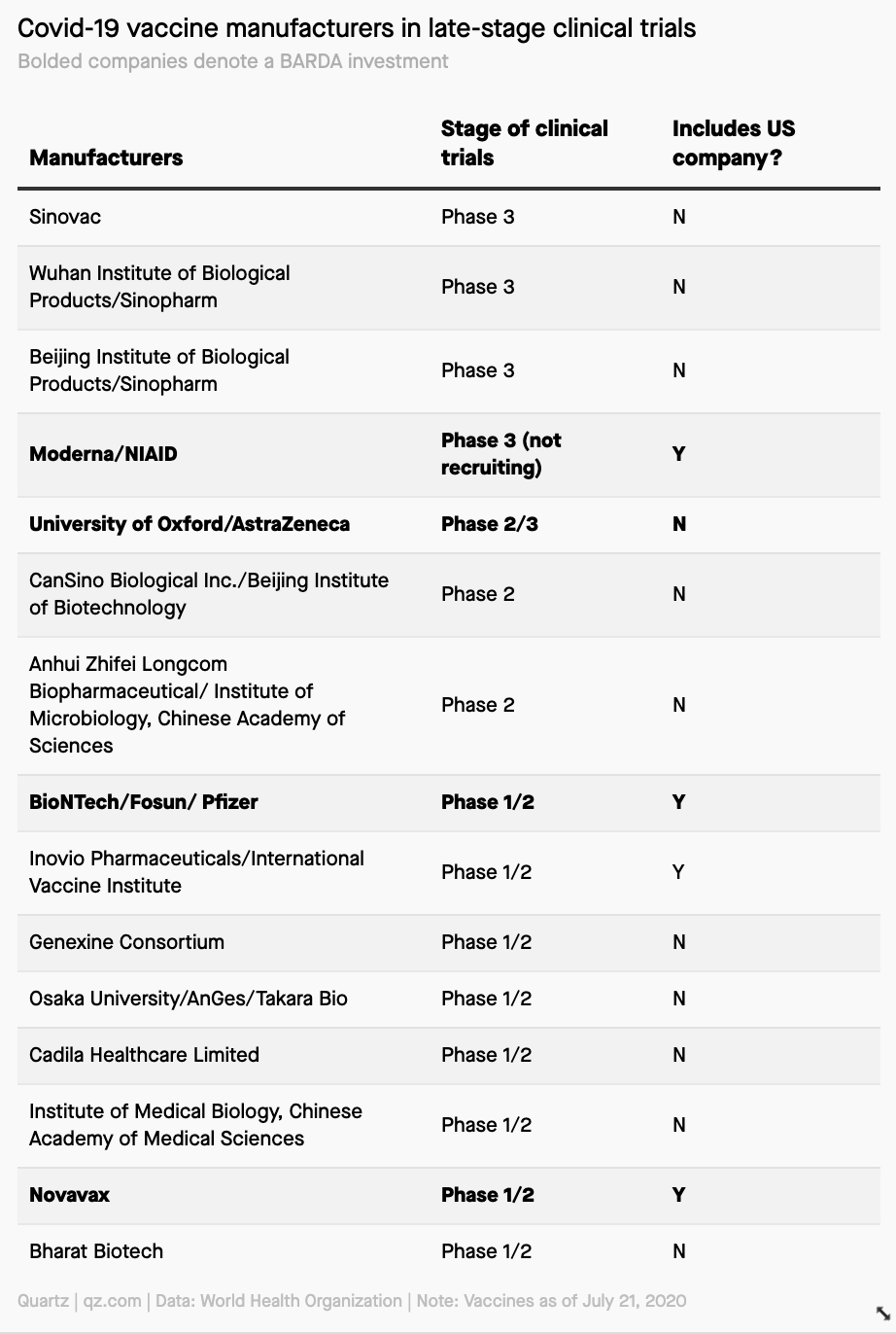
A couple of notes:
Clinical trials have three phases: 1, 2, and 3. Phase 3 is the last trial a drug or vaccine must pass before gaining regulatory approval.
The bolded companies are those that have received grants from the US government to buy pre-emptive vaccines, assuming their clinical trials continue to go successfully.
The US has not invested in three of the five vaccines that are in Phase 3 trials.
Bonus fact: One man voices all your favorite cartoon characters.
I’ve needed to find happy outlets to recharge. For me, that’s been Avatar: The Last Airbender. (Yes, it’s a cartoon children’s show—just trust me on this one.)
One day while watching the credits, I saw they credited the voice actor who plays Appa and Momo…who are both animals on the show, and make only animal noises.

Appa, a flying white bison (I think he’s a beaver-bison, but they only ever refer to him as a Sky Bison).
Turns out the guy who voices Appa and Momo, Dee Bradley Baker, is an animal voice actor extraordinaire. He’s also the voice behind Lion in Steven Universe and Mandu in Kipo and the Age of Wonderbeasts. He’s got some 600+ voice acting credits, including appearances on shows like American Dad and Family Guy. Can you even imagine having that job? Just to make fictional animal voices? And then, be one of the best in the world at it? Incredible!
Super bonus fact: Need some more funny distraction? Check out this work by Victoria Edel in the New Yorker’s Daily Shouts.
Also, subscribe to her newsletter.
That’s all for now—stay curious, friend ❤️
If you love Scrap Facts, consider hitting the “like” button at the bottom of this page, or sending it to a friend. You can also send your own scrap facts to scrapfacts@gmail.com to be featured in future editions. Wanna keep in touch outside of this newsletter? Follow me on Twitter and Instagram.

Top image by E. Y. Smith, headshot drawing by Richard Howard.


